Artigo
| Oxidation of cyclohexanol on phosphotungstic acid anion intercalated layered double hydroxides with aqueous H2O2 as oxidant |
|
Xueli BaiI; Zhaoyang BaiII; Dandan XueI; Huiyan SunI; Xin HuangI; Yongxiang ZhaoI,#; Yue ZhangI,*
IEngineering Research Center of Ministry of Education for Fine Chemicals, School of Chemical Engineering, Shanxi University, Taiyuan 030006, Shanxi, China Recebido em 03/06/2017 *e-mail: zyue@sxu.edu.cn The layered double hydroxides (LDH) of Mg2AlNi and Mg3Al pillared by Keggin-type phosphotungstic acid anion (POM), i.e. Mg2AlNi-POM LDH and Mg3Al-POM LDH were synthesized by an ion-exchange method. The synthesized POM intercalated LDH compounds were characterized using various techniques such as FTIR, XRD, TGA and BET. The observed results show that the obtained catalysts retain the layer structure of LDH. Compared with the binary Mg3Al-POM LDH, the ternary Mg2AlNi-POM LDH catalyst indicated a higher thermal and chemical stability. The catalytic activity of the resulting LDH-POM was also assessed in the green oxidation of cyclohexanol with aqueous H2O2 as an oxidant. The Mg2AlNi-POM LDH showed a much higher conversion and selectivity for cyclohexanone than the corresponding Mg3Al-POM LDH catalyst. INTRODUCTION It is well known that the carbonyl compounds have widespread applications in dyestuff, perfumery and agro chemical industries.1-3 Oxidation of alcohols has traditionally been the major route for the preparation of carbonyl compounds. However, many of the traditional inorganic oxidants utilized for the oxidation of alcohols are hazardous or toxic, for example Na2Cr2O7, NaClO4 and KMnO4. Therefore, it is necessary to find green and clean catalysts and oxidants for the oxidation of alcohols. One ideal oxidant for the oxidation of alcohols is hydrogen peroxide as it can oxidize the organic compounds with the formation of water as a by-product.4,5 Noyori reported various polyoxometalate (POM) catalysts in the oxidation of organic compounds with aqueous H2O2 as the oxidant. (POMs)6-9 have widely been used in the catalytic oxidation reaction due to their oxidative stability and high efficiency.10-12 The homogeneous catalysis is green, highly efficient and environmentally pollution-free,13,14 but there are inherent problems existing with this method such as the difficult separation of the product and thus the catalyst is easily lost in the process. Hence, it is absolutely necessary to heterogenize the soluble POMs. In this context, one potential strategy could be by using the layered double hydroxides (LDHs), which are the type of anionic clay materials with layered structure. The layers as a result carry positive charges that are balanced by the interlayer anions15 which could be replaced by various POM anions through ion-exchange.15-20 This provids an effective route for heterogenizing the catalysts in a homogeneous medium. Besides, these POM-pillared LDH (POM/LDH) materials were tested for the oxidation of benzaldehyde,21 cyclohexene22 and allyl alcohol23 with aqueous H2O2 in the absence of an organic solvent. The observed results indicate that LDH precursors can effectively influence the conversion and selectivity in the catalytic reaction. In addition, POM/LDH showed a high recovery rate and good thermal stability24 in the catalytic process. The diversity of POM/LDH is due to the adjustable oxyacid of POM as well as adjustable distribution of the metal cations on the surface of LDH layers, which lead to a difference in the structure and catalytic performance. To date, the studies related to POM/LDH mainly focusing on changing the composition of POMs, and their LDH precursors largely utilizing binary Mg/Al-LDH or Zn/Al-LDH (for instance, Mg2Al-W7O246- LDHs,25 Zn2Al-[SiW11O39]0.12·0.2H2O LDH15 and ZnAl-[EuW10O36]9-/ [Eu(BW11O39)(H2O)3]6-/[Eu(PW11O39)2]11- LDH26 etc.). However, the Mg/Al-LDH precursor easily causes a severe acid-base reaction with the POM solution.23-27 The reaction can be controlled through changing the composition of LDH precursor. In recent years, non-precious metals such as Ni,28,29 V30 and Cu31,32 have been successfully used as heterogeneous catalysts in modifying the inorganic matrices as for the oxidation of alcohols. Nickel is usually applied as an active species for the reduction reaction. In the current work, we reduced the basicity of LDH precursor by adding nickel to the binary Mg/Al-LDH, as the basicity of nickel is weaker than magnesium. The ternary catalyst has good stability. Moreover, it is well known that nickel is usually applied as an active species in the heterogeneous catalysis for the oxidation of alcohol.28,29 Therefore, the performance could be improved in the oxidation of cyclohexanol with aqueous H2O2 as an oxidant by adding a metal nickel to the binary Mg/Al-LDH. We hereby reported the preparation and characterization of ternary Mg2AlNi-POM LDH catalyst using 12-phosphotungstic acid anions ([PW12O40]3-, referred to as "POM") which are intercalated between the LDH layers replacing the nitrate anions via ion-exchange pathway. Also, the structure of Mg2AlNi-POM LDH and its catalytic performance in the oxidation of cyclohexanol have been compared with the Mg3Al-POM LDH catalyst. As the precursor, the ternary Mg2AlNi-LDH not only improves the stability of POM-LDH catalyst but also enhances the selectivity for the oxidation of cyclohexanol under mild conditions and in presence of water as solvent.
EXPERIMENTAL Materials and methods Mg(NO3)2·6H2O(AR), Al(NO3)3·9H2O (AR) and Ni(NO3)2·6H2O (AR) were purchased from the Tianjin Hengxing Chemical manufacturing co., LTD. Na3[PW12O40] (AR) and cyclohexanol (AR) were purchased from Sinopharm Chemical manufacturing co., LTD. Aqueous H2O2 (30 wt%; AR) was purchased from Beijing chemical factory. X-ray diffraction (XRD) patterns were recorded on a German Bruker D8 Advance X-ray powder diffraction system (CuKα, 40 kV, 40 mA). FT-IR spectra were collected on a Bruker Tensor 27 infrared spectrometer, using the KBr pallet method. TEM micrographs were taken using Japanese Jeol TEM-2100 type transmission electron microscopy. Gas chromatography(GC) were used Agilent Technologies GC-7890B type chromatograph, N2 as carrier gas. The nitrogen sorption experiments were performed at 196 ºC on an ASAP 2020 system. TGA/DTA analysis using STA449F3 type comprehensive thermal analyzer was produced of Germany NETZSCH, the heating rate of 10 ºC/ min. The crucible material is alumina. The gas is a mixture of nitrogen and oxygen and their rate is 5mL/min and 20mL/min respectively. Preparation of catalyst precursors and LDH-POM catalysts A solution containing 30 mmol of Mg(NO3)2·6H2O and 10 mmol of Al(NO3)3·9H2O in 100 mL of decarbonated water and an aqueous solution of 1mol/L NaOH were simultaneously added dropwise to a 250 mL three-necked flask having (with 50 mL of deionized water) which was under with rapid stirring and under nitrogen atmosphere. The relative rates of adding the mixed solution of nitrate salts and the 1 mol/L of NaOH solution were adjusted to maintain the pH ≈ 10. The resulting suspension was kept at 80 ºC for 12 h in the hydrothermal synthesis reactor. Then, the slurry was separated into two equal portions. One portion was centrifuged and washed with water for several times (until pH ≈ 7), and the resulting precipitate was dried at 120 ºC overnight to obtain Mg3Al-NO3 LDH which was used for further characterization. After centrifugation and washing, the other portion of the precipitate was dispersed in 100 mL water and maintained under N2 as Mg3Al-NO3 LDH slurry which was simultaneously added dropwise with POM (10 mmol) to a 250 mL three-necked flask. The mixed slurry was stirred for 3 h at 60 ºC. The pH of the dispersion was around 4.8, which is suitable for the POM anion as the [PW12O40]3- is stable in the pH range of 2-6.33 Finally, Mg3Al-POM LDH catalyst was obtained. The synthesis of Mg2AlNi-NO3 LDH and Mg2AlNi-POM LDH slurry is similar to Mg3Al-NO3 LDH and Mg3Al-POM LDH analogue which were obtained using Mg(NO3)6·6H2O (20mmol), Al(NO3)3·9H2O (10 mmol) and Ni(NO3)2·6H2O (10 mmol). In addition, we prepared a series of POM intercalated materials i.e. Mg/Al/Ni-POM LDH using the same method as above. The molar ratio of the LDH material i.e. Mg+Ni/Al is of 3:1 and the molar ratio of POM (1.43 mmol, 1.43×7 ≈10 mmol) to the exchange negative charge capacity (10 mmol) of LDH is 1:7. Oxidation of cyclohexanol In a typical experiment, a mixture of cyclohexanol (10 mmol), ethanol (100 mmol), aqueous H2O2 (30%, 5 mmol) and 0.2 g of catalyst were refluxed at 90 ºC for 2 h. The resulted mixture was then cooled and toluene (10 mmol, internal standard) was added to the liquid phase and was then analyzed by GC to determine the conversion and selectivity. After being washed with ethanol and dried under vacuum, the recovered catalyst could be reused.
RESULTS AND DISCUSSION Synthesis and characterization of LDH-POM catalysts LDH-POM catalysts were synthesized through a synthetic route where POM anion could be directly inserted into LDH layers without adjusting the pH. XRD patterns of the as-prepared samples have been represented in Figure 1, and the major d(003) spacings (calculated using Bragg's equation, λ=2dsinθ, where λ=0.15406 nm) are given in Table 1. Mg2AlNi-POM LDH and Mg3Al-POM LDH have lower diffraction intensity of peaks compared with Mg2AlNi-NO3 LDH and Mg2AlNi-NO3 LDH. Figure 1 shows that the peak position of POM/LDH moves to lower 2-theta -diffraction angle, owing to the intercalation of POM anion intercalated into LDH. Furthermore, the major d(003) spacings of Mg2AlNi-POM LDH and Mg3Al-POM LDH demonstrate a higher value. The d(003) spacing of LDH-POM includes the thickness of LDH host and the gallery height of interlayer region. Subtracting the thickness of host layer which is assumed to be about 0.48 nm gives the gallery heights of the LDH-POM to be about 0.98~1.01 nm. The observed gallery heights are in agreement with the diameter of the short axis of POM anions (~0.98nm).33 The major diffraction peak and a low intensity peak, is indicated as (003). This mixture includes POM-intercalated LDH and the POM-immobilized LDH (POM anions immobilized on the external surface of LDH layers). Besides, the diffraction peak (110) is detected in all the samples which indicates that the layered structure of LDH is retained after immobilization. The intensity of the diffraction peak of (110) plane of POM-LDH is low, especially the Mg3Al-POM LDH, which may be due to acid-base reaction that occurs between the acidic POM solution and the basic Mg3Al-NO3 resulting in a partial decomposition of LDH host.
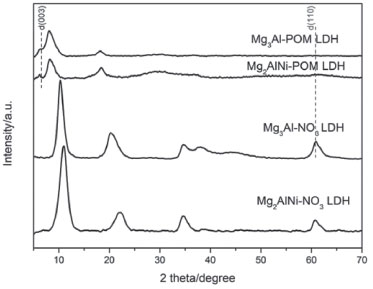 Figure 1. XRD patterns of different LDH-NO3 supports and LDH-POM catalysts
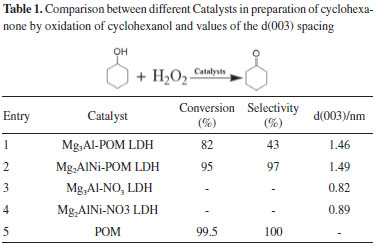
These results suggest that the POM anion has been successfully incorporate into the interlayer of LDH by the anion-exchange process. The FT-IR spectra of the samples are shown in Figure 2. After intercalation, Mg2AlNi-POM LDH and Mg3Al-POM LDH showed the characteristic peaks at 812 and 742 cm-1, which are related to the W-Oc-W and W-Oe-W (c, corner; e, edge sharing) vibrations. The bands at 958 and 902 cm-1 are attributed to the vibration of W-Ot (t, terminal), and the peaks at 1083 and 1058 cm-1 are attributed to that of P-O. The Mg2AlNi-NO3 LDH and Mg2AlNi-NO3 LDH show a strong peak at 1384 cm-1 due to the vibration of N-O from nitrate anion in the interlayer region.33 However, a weak peak of N-O still appeared from Mg2AlNi-POM LDH and Mg3Al-POM LDH. These results suggest that the POM anions were exchanged into LDH which is in agreement with the results obtained from XRD patterns. However, based on the band appearing at 1384 cm-1 and the band at 1365 cm-1 which are attributed to the C-O bonds, suggesting that the nitrate and the carbonate anions are incorporated between the layers of LDH. It illustrates that the anion exchange reaction is not completed and some of the nitrate anions remained partly and some samples are contaminated with carbonate anion.
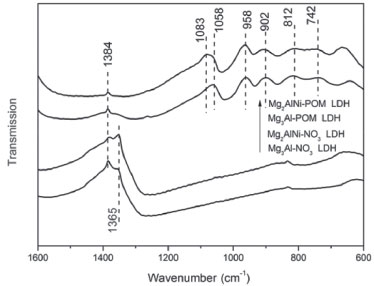 Figure 2. FT-IR spectra of different LDH-NO3 and LDH-POM samples
According to the TEM images from Figure 3. Mg2AlNi-POM LDH and Mg3Al-POM LDH keep the plate morphology for LDH host. Compared with Mg2AlNi-NO3 LDH and Mg2AlNi-NO3 LDH, LDH host of Mg3Al-POM LDH is not clearly observed because of the partial decomposition of LDH host in the anion-exchange process.23 Furthermore, the layer of Mg2AlNi-POM LDH is more complete than Mg3Al-POM LDH which maybe due to the addition of nickel species reducing the alkalinity of LDH hosts to effectively restrain the decomposition of LDH host.
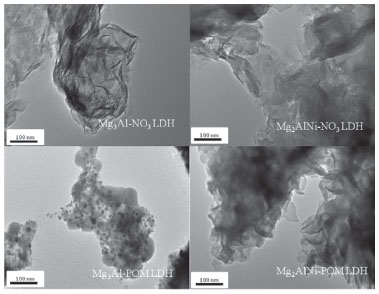 Figure 3. TEM images of different LDH-NO3 and LDH-POM samples
The thermal stability of samples were studied using TGA and the obtained results have been shown in Figure 4. The values of the major losses produced during LDH decomposition are shown in Table 1. The first weight loss (peak 1) is broader and appearing clearly at temperatures below 200 ºC, which could be attributed to the loss of non-structural water physisorbed on the external surface of the LDH crystallites. From 200 to 500 ºC (peak 2), the observed weight losses could be associated to water differently bonded to the structure such as interlayer water co-intercalated with nitrate or chemisorbed water.34 Ternary Mg2AlNi-POM LDH and Mg2AlNi-NO3 LDH are more stable than binary Mg3Al-POM LDH and Mg2AlNi-NO3 LDH.
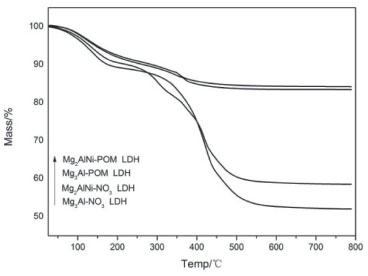 Figure 4. TGA traces for the representative LDH-NO3 and LDH-POM samples
Catalytic performance of Mg2AlNi-POM LDH in the oxidation of cyclohexanol Catalytic performance between Mg2AlNi-POM LDH and Mg3Al-POM LDH is different for the oxidation of cyclohexanol (Table 1). Although POM has good catalytic performance, it is a homogeneous catalysis. Mg2AlNi-POM LDH exhibits higher conversion and selectivity to cyclohexanone (the catalytic activity of Mg2AlNi-POM LDH was evaluated in triplicates and the error bars have been shown in Figure 5) than Mg3Al-POM LDH, which is due to the synergistic effect of nickel. This is further proved through the results as shown in Table 2 which indicates the comparison between catalysts with different molar ratio of Ni in the preparation of cyclohexanone by the oxidation of cyclohexanol. We could clearly conclude that the conversion and selectivity to cyclohexanone increase with an increase in the molar ratio of Ni of POM-LDH.
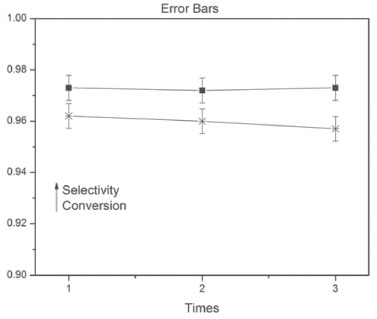 Figure 5. Error bars on selectivity and conversion of Mg2AlNi-POM LDH catalysts
In addition, the decomposition of hydrogen peroxide can also affect the catalytic performance. When pH is less than 3.5, hydrogen peroxide maintains the stability.35 The measured pH of the catalytic environment of Mg3Al-POM LDH and Mg2AlNi-POM LDH were 4.7 and 4.3, respectively. It exhibits that the addition of nickel could reduce the alkalinity of the material itself and reduces the rate of decomposition of hydrogen peroxide. Stability and recyclability of LDH-POM catalysts The LDH-POM catalysts could be reused with negligible loss of catalytic activity after five runs (Figure 6). The cyclohexanone's selectivity retained over 95% even after its usage for five times. However, the activity of Mg3Al-POM LDH catalyst is decreased after the first reuse, suggesting that the Mg3Al-POM LDH is unstable towards the oxidation of cyclohexanol. These results demonstrate that Mg2AlNi-POM LDH is a stable heterogeneous catalysis in the aqueous oxidation. The recovered Mg2AlNi-POM LDH catalyst after the fifth cycle was characterized again by XRD (Figure 7) and FT-IR (Figure 8) to evaluate the structural stability. Compared to the fresh catalyst, no significant changes were observed in the XRD and IR spectra. These observations provide direct evidence that the Mg2AlNi-POM LDH catalyst is stable under the subjected reaction conditions.
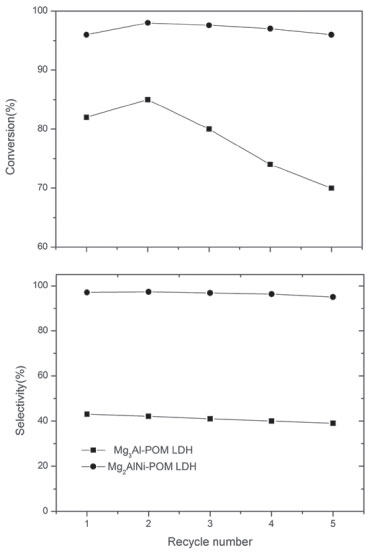 Figure 6. Recyclability of Mg3Al-POM LDH and Mg2AlNi-POM LDH catalysts
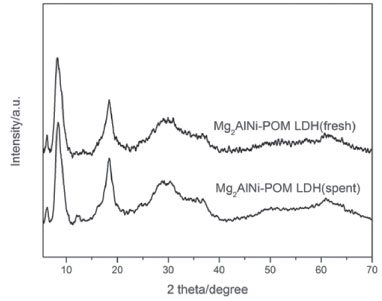 Figure 7. XRD patterns of fresh Mg2AlNi-POM LDH and spent Mg2AlNi-POM LDH
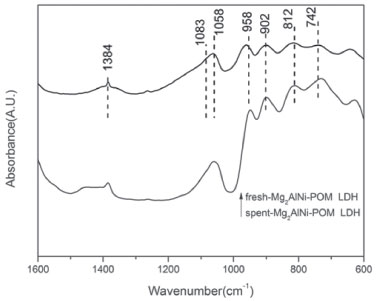 Figure 8. FT-IR spectra of fresh Mg2AlNi-POM LDH and spent Mg2AlNi-POM LDH
CONCLUSIONS In summary, we have fabricated ternary catalysts through ion-exchange method without adjusting the pH. The followed steps for the intercalation of POM anion into LDH is convenient, environmentally benign and more importantly cheaper. Besides, it provides an efficient heterogeneous oxidation catalysis. Furthermore, we have examined the catalytic performance of Mg2AlNi-POM LDH in the oxidation of cyclohexanol as compared with the conventional catalyst, Mg3Al-POM LDH. The resulting heterogeneous catalysis Mg2AlNi-POM LDH showed a much higher catalytic performance than the corresponding catalyst i.e. Mg3Al-POM LDH. This is mainly due to the synergetic effects between nickel and POMs. The original crystal structure of Mg2AlNi-POM LDH was maintained. Additionally, the Mg2AlNi-NO3 LDH precursor with lower surface basicity is more effective than the Mg3Al-NO3 LDH, keeping the stability of the material and reducing the hydrolysis of aqueous H2O2. Overall, this work offers a green chemistry strategy for the improvement of heterogeneous oxidation using aqueous H2O2.
ACKNOWLEDGMENTS This work was supported by the National Natural Science Foundation. (NO. 20573071)
REFERENCES 1. Pybus, D. H. In The chemistry of fragrances; Pybus, D. H.; Sell, C. S., eds.; Royal Society of Chemistry: London, 2006. 2. Singh, R. P.; Subbarao, H. N.; Dev, S.; Tetrahedron 1979, 35, 1789. 3. Fey, T.; Fischer, H.; Bachmann, S.; J. Org. Chem. 2001, 66, 8154. 4. Ishii, Y.; Yamawaki, K.; Ura, T.; J. Org. Chem. 1989, 20, 3587. 5. Noyori, R.; Aoki, M.; Sato, K.; Chem. Commun. 2003, 34, 1977. 6. Kortz, U.; Müller, A.; Slageren, J. V.; Coord. Chem. Rev. 2009, 253, 2315. 7. Keita, B.; Liu, T.; Nadjo, L.; ChemInform 2009, 40, 19. 8. Bi, L. H.; Hou, G. F.; Li, B.; Wu, L. X.; Kortz, U.; Dalton Trans. 2009, 32, 6345. 9. Long, D. L.; Tsunashima, R.; Cronin, L.; Angew. Chem., Int. Ed. 2010, 49, 1736. 10. Grigoropoulou, G.; Clark, J. H.; Elings, J. A.; Green Chem. 2003, 5, 1. 11. Lane, B. S.; Burgess, K.; Chem. Rev. 2003, 103, 2457. 12. Mizuno, N.; Yamaguchi, K.; Kamata, K.; J. Coord. Chem. Rev. 2005, 249, 1944. 13. Mizuno, N.; Kamata, K.; Yamaguchi, K.; Top. Catal. 2010, 53, 876. 14. Lv, H.; Geletii, Y. V.; Zhao, C.; Chem. Soc. Rev. 2012, 41, 7572. 15. Guo, Y.; Li, D.; Hu, C.; Microporous Mesoporous Mater. 2002, 56, 153. 16. Guo, Y.; Li, D.; Hu, C.; Int. J. Inorg. Mater. 2001, 3, 347. 17. Guo, Y.; Li, D.; Hu, C.; Appl. Catal., B 2001, 30, 337. 18. Pinnavaia, T. J.; Kwon, T.; Dimotakis, E. D.; US pat. 5079203, 1992. 19. Nijs, H.; Bock, M. D.; Maes, N.; Porous Mater. 1999, 6, 307. 20. Hasannia, S.; Yadollahi, B.; Polyhedron 2015, 99, 260. 21. Wei, X.; Fu, Y.; Xu, L.; J. Solid State Chem. 2008, 181, 1292. 22. Franklin, I. L.; Ph. D. Thesis, University of London, London 2005. 23. Liu, P.; Wang, C.; Li, C.; J. Catal. 2009, 262, 159. 24. Guo, J.; Sun, T.; Shen, J.; Acta Sci. Nat. Univ. Jilin. 1995, 16, 346. 25. Arco, M. D.; Carriazo, D.; Gutiérrez, S.; Inorg. Chem. 2004, 43, 375. 26. Sousa, F. L.; Ferreira, A. S.; Ferreira, R. A. S.; ChemInform 2006, 37, 726. 27. Jana, S. K.; Kubota, Y.; Tatsumi, T.; J. Catal. 2008, 255, 40. 28. Choudary, B. M.; Kantam, M. L.; Rahman, A.; Angew. Chem., Int. Ed. 2001, 40, 763. 29. Choudhary, V. R.; Chaudhari, P. A.; Narkhede, V. S.; Catal. Commun. 2003, 4, 171. 30. Velusamy, S.; Punniyamurthy, T.; Org. Lett. 2004, 6, 217. 31. Meng, X.; Lin, K.; Yang, X.; J. Catal. 2003, 218, 460. 32. Meng, X. J.; Xiao, F. S.; Acta Phys.-Chim. Sin. 2004, 20, 939. 33. Liu, Y.; Murata, K.; Hanaoka, T.; J. Catal. 2007, 248, 277. 34. Guil-López, R.; Navarro, R. M.; Peña, M. A.; Int. J. Hydrogen Energy. 2011, 36, 1512. 35. Zhang, Q.; Ying, C. Y.; Ke-Na, Y. U.; Journal of Jiaxing University 2010. |
On-line version ISSN 1678-7064 Printed version ISSN 0100-4042
Qu�mica Nova
Publica��es da Sociedade Brasileira de Qu�mica
Caixa Postal: 26037
05513-970 S�o Paulo - SP
Tel/Fax: +55.11.3032.2299/+55.11.3814.3602
Free access





